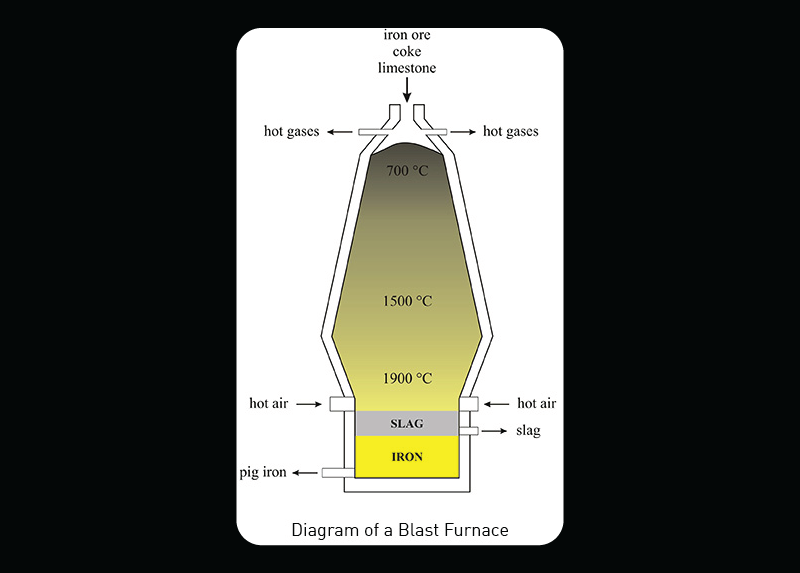
Blast furnaces are used to produce iron. Iron production is one of America’s backbone industries. Iron was the material that gave rise to the Industrial Revolution. Abraham Darby created the first blast furnaces fueled with coke instead of charcoal. Charcoal is created from burning trees and requires a lot of labor and resources. Coke is created from coal which is less labor intensive and saves community timber resources. The coke mixed with iron ore and limestone created a much stronger iron than could be made with charcoal.
Modern blast furnaces are made from refractory brick surrounded by steel. The hollow stacks receive raw iron ore, coke, and limestone through the opening at the top. Hot air is blown into the bottom of the stack. The materials melt and combine to form liquid iron and slag which drains at the bottom. The combination of high heat with the chemical compounds found in each raw material cause chemical reactions to occur within the furnace. These chemical reactions purify the iron ore and create iron. The chemical reactions and the temperatures they occur are listed below.
| 1) 3 Fe2O3 + CO = CO2 + 2 Fe3O4 | Begins at 850° F |
| 2) Fe3O4 + CO = CO2 + 3 FeO | Begins at 1100° F |
| 3) FeO + CO = CO2 + Fe or FeO + C = CO + Fe |
Begins at 1300° F |